In this issue
- Biosimilars: The next generation of biologic treatment for inflammnatory arthritis patients
- What are biologic biosimilars?
- Biosimilars in Canada—approved and under review
- What does transitioning mean?
- The biosimilar transitioning experience
- Biosimilar Transitioning: The Canadian experience
- Biosimilars in Canada—What inflammatory arthritis patients need to know
- Societal benefits of biosimilars
JointHealth™ insight Published June 2019

Provincial and private drug benefit plans have begun implementing policy transition requiring patients to move from their current biologic originator to its biologic biosimilar. Policy transition has been successfully implemented in many European countries and in thousands of inflammatory arthritis patients with no compromise to patient safety, effectiveness or quality of care.
On May 27, 2019, British Columbia became the first jurisdiction in North America to introduce biosimilar policy transition with the launch of the Biosimilars Initiative, which will implement new reimbursement coverage for patients on infliximab or etanercept for rheumatoid arthritis, ankylosing spondylitis, and psoriatic arthritis.
Between May 27, 2019 and November 22, 2019, B.C. patients on Remicade® will be transitioned to its biosimilar, either infliximab-Inflectra® or infliximab-Renflexis®. Patients on Enbrel® will be transitioned to its biosimilar, either etanercept-Brenzys® or etanercept-Erelzi®. In each case, patients and their rheumatologist will be able to choose between the two available biosimilars.
Over the six month period, reimbursement coverage will be provided for all originator and biosimilar brands, in order to provide time for patients to speak with their rheumatologist about transitioning to a biosimilar.
Biosimilars are next generation biologic medications that have been approved for use in Canada since 2009, and in arthritis, since 2014. Fourteen biosimilars are currently approved by Health Canada with another 15 under review. Since the European Union (EU) approved the first biosimilar in 2006, the EU has approved 40 biosimilars.
In this special issue of JointHealth™ insight, we provide patients with inflammatory arthritis important biosimilars facts to help them understand the principles of this newest type of biologic and have an informed conversation with their healthcare provider.
What are biologic biosimilars?
 Biologics are life-saving “advanced therapy” treatments – meaning they are used after conventional disease modifying anti-rheumatic drugs (csDMARDs), such as methotrexate, hydroxychloroquine and sulfasalazine – that can help patients with inflammatory forms of arthritis. They are made from complex large molecules that come from living cells. Biologics are time consuming to research, develop and manufacture, making them expensive when they first come into the market place as a patented medicine. This can limit patients’ reimbursement access to such medicines and can make it difficult for the health care system to afford them.
Biologics are life-saving “advanced therapy” treatments – meaning they are used after conventional disease modifying anti-rheumatic drugs (csDMARDs), such as methotrexate, hydroxychloroquine and sulfasalazine – that can help patients with inflammatory forms of arthritis. They are made from complex large molecules that come from living cells. Biologics are time consuming to research, develop and manufacture, making them expensive when they first come into the market place as a patented medicine. This can limit patients’ reimbursement access to such medicines and can make it difficult for the health care system to afford them.
Biosimilars are the next generation of biologic treatment and have nearly the same effectiveness, safety, immunogenicity profile and quality and deliver the same therapeutic benefits to patients as their originator; in some studies, slightly better.
After meeting Health Canada’s strict safety and efficacy requirements, biosimilars enter the market after a biologic originator patent expires, just like a small molecule generic medication does when the brand name medication’s patent expires. And just as generic medications are much lower in price compared to their original versions, so too are biosimilars.
For example:
Biosimilars have been approved for use in Canada since 2009, and in inflammatory arthritis since 2014 – there are more biologic originators and biosimilars in use in inflammatory arthritis than in any other disease.
Since the European Union (EU) approved the first biosimilar in 2006, it has approved the highest number of biosimilars worldwide, gaining experience of their use and safety. During this period, the EU monitoring system for safety concerns has not identified any relevant difference in the nature, severity or frequency of adverse effects between biosimilars and their biologic originators.
Biosimilars in Canada – approved and under review
What does transitioning mean?
Will transitioned patients receive the same level of patient support from biosimilar manufacturers?
Patients should expect the same or similar types of support from all biologic patient support programs, no matter who makes their medication. The rheumatologist will give their patient the name of the patient support program coordinator for their biosimilar and they will help organize reimbursement and assist with other patient needs. The patient and their rheumatologist will monitor the safety and effectiveness of the biosimilar as part of routine patient care.
There are two types of transitions
The biosimilar transitioning experience
The leading regulators in the world – including the European Medicines Agency, Food & Drug Administration in the U.S and Health Canada – support well-controlled transition.
Patients need to know transition policy has been safely and effectively implemented over the past 10 years with thousands of patients with autoimmune diseases such as rheumatoid arthritis, psoriatic arthritis, gastrointestinal and bowel diseases in many countries in Europe with no compromise to patient safety, effectiveness or quality of care.
 More than 100 research studies exist looking at patients who have successfully policy transitioned from a TNF inhibitor biologic originator to its TNF inhibitor biologic biosimilar and show no health differences between patients.
More than 100 research studies exist looking at patients who have successfully policy transitioned from a TNF inhibitor biologic originator to its TNF inhibitor biologic biosimilar and show no health differences between patients.
Today, the international inflammatory arthritis community has the most biosimilar experience of any disease community, with tens of thousands of patients safely and effectively transitioned to a biosimilar.
Biosimilar Transitioning: The Canadian experience
In Canada, federal, provincial, territorial and private insurance drug plans have begun implementing policy transition, which moves patients from their current biologic originator to its biologic biosimilar. The European experience of rheumatologists successfully working with their patients to transition them from an originator to a biosimilar has helped inform policy transition development in Canada.
On May 27, 2019, British Columbia became the first jurisdiction in North America to introduce biosimilar policy transition with the launch of the Biosimilars Initiative. According to the Ministry of Health, the goal of the Biosimilars Inititiave is to improve the sustainability of its PharmaCare program by expanding the use of biosimilars, which will create opportunities for new drug listings and boost existing coverage for patients.
Between May 27, 2019 and November 22, 2019, approximately 240,000 B.C. patients living with either ankylosing spondylitis, diabetes, plaque psoriasis, psoriatic arthritis or rheumatoid arthritis will have six months to work with their health-care team to transition their prescription from the biologic to the biosimilar drug by Nov. 25, 2019. After that time, PharmaCare will no longer provide coverage for the original biologic drug for these indications, except for exceptional cases.
An immediate effect of this announcement is the listing on BC PharmaCare of the psoriatic arthritis medication ixekizumab (Taltz®) as well as the diabetes medication empagliflozin (JARDIANCE™).
“B.C. is leading the country by promoting the widespread use of biosimilars, which have been proven to work just as safely and effectively as higher priced biologics. To date, Canada is far behind European jurisdictions,” said Adrian Dix, Minister of Health. “Biosimilars are a necessary step to ensure PharmaCare provides existing coverage for more people and funds new drugs well into the future.”
The Ministry of Health further explained that despite clear evidence to support the use of biosimilars – highly similar versions of biologics that cost anywhere from 25% to 50% less – Canada has a significantly lower use rate for biosimilars compared to other jurisdictions, at approximately 8%. Other nations have upwards of 50 to 95%.
Biologics, collectively, are the single biggest expense for public drug plans and their costs continue to climb. In 2018, B.C. spent $125 million on Lantus, Enbrel and Remicade, three biologic medications that treat chronic diseases such as diabetes, arthritis and Crohn’s disease.
“We know that our use rates can be much higher. For example, the biosimilars for infliximab (known as Inflectra® and Remsima®) have combined 5 to 10% use rate in Canada after four years on the market, compared to Norway (98%), United Kingdom (90%) and Scotland (85%). As a result, in 2017, Canada spent over $1.1 billion on the original biologic drug called Remicade®,” added Minister Dix.

Having an evidence-based biosimilar transition conversation with your healthcare professional
Canadian regulators recommend, prior to transition, a discussion occurs between a health care provider and their patient.
The most important element for patients going through a transition is access to patient-friendly, science-based information to support their conversations with their health care team.
Arthritis Consumer Experts (ACE) has been a leader in biosimilars discussions since 2009, sharing information with stakeholders across Canada through free research-based workshops, webinars and education programs. Drawing from this experience, ACE has created the Biosim•Exchange, an information hub for consumers to get the latest biosimilars news and background analysis. http://bit.ly/BiosimExchangeEN.
ACE has produced a special biosimilars education video series, where ACE speaks to medical experts on key patient questions around biosimilars and transitioning from originators to biosimilars: Go to https://biosim.jointhealth.org/resources and click on “Biosimilars education videos.”
ACE has also recently published a new guide -
Societal benefits of biosimilars
Why societal benefits are important to patients
Evidence-based private or public drug plan transition policy could help more patients (not just those to have been lucky enough to receive coverage for an originator under current and often restrictive special access criteria), the healthcare system, and society.
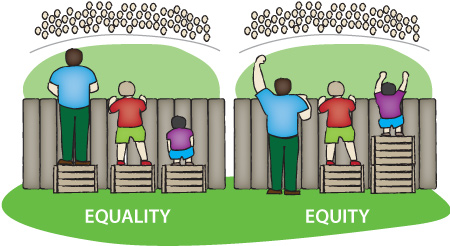
Societal benefit from biosimilar transition policy is not a “pipe dream” – it is a real opportunity to help more patients get on costeffective therapies that are also “state-of-the-art” in the treatment of inflammatory arthritis and other autoimmune diseases driven by inflammation.
Societal benefits to taxpayers
A key benefit of transitioning patients is the hundreds of millions of dollars in cost savings to the health care system.
In a presentation at a CADTH conference in April 2019, Elena Lungu from the Patented Medicine Prices Review Board (PMPRB) estimated the potential missed biosimilars savings in Canada for 2019 (versus the Organisation for Economic Cooperation and Development (OECD) countries with the highest biosimilars uptake are achieving):
That translates into a total of $791 million of biosimilars savings in 2019 potentially not realized by Canadian public and private drug plans.
A study presented at the 2019 Canadian Rheumatology Association annual meeting looked at biosimilar use and cost in Canada under provincial drug plans in 2016 (excluding Quebec) and found that if half of the Remicade claims has been Inflectra instead, the cost difference would have been more than $102 million1.
According to a 2018 study commissioned by the PMPRB and conducted by the National Prescription Drug Utilization Information System (NPDUIS), “Potential Savings from Biosimilars in Canada”, estimated potential annual savings for a single biologic originator, like infliximab, in Canada could range between $91M and $514M in the third year following biosimilar entry.
Evidence-based provincial drug plan and health insurer transition policy could help more patients, the healthcare system, and society, if savings from biosimilars use was redirected to:
1. Moura CS, Choquette D, Biore G, Bykerk VP, Thorne C, Maksymowych WP, Lakatos P, Bessissow T, Svenson L, Targownik L, Afif W, Bernatsky S. Inflectra and Remicade Use and Cost in Canada Under
Provincial Drug Plans in 2016 [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 10).
Listening to you
We hope you find this information of use. Please tell us what you think by writing to us or emailing us at feedback@jointhealth.org. Through your ongoing and active participation, ACE can make its work more relevant to all Canadians living with arthritis.
Update your email or postal address
Please let us know of any changes by contacting ACE at feedback@jointhealth.org. This will ensure that you continue to receive your free email or print copy of JointHealth™ insight.
Arthritis Consumer Experts (ACE)
Who We Are
Arthritis Consumer Experts (ACE) operates as a non-profit and provides free research based education and information to Canadians with arthritis. We help (em)power people living with all forms of arthritis to take control of their disease and to take action in healthcare and research decision making. ACE activities are guided by its members and led by people with arthritis, scientific and medical experts on the ACE Advisory Board. To learn more about ACE, visit www.jointhealth.org
Disclosures
Over the past 12 months, ACE received grants-in-aid from: Amgen, Arthritis Research Canada, Canadian Biosimilars Forum, Canadian Institutes of Health Research, Canadian Rheumatology Association, Eli Lilly Canada, Hoffman-La Roche Canada Ltd., Knowledge Translation Canada, Merck Canada, Novartis Canada, Pfizer Canada, Sandoz Canada, Sanofi Canada, UCB Canada, and the University of British Columbia.
ACE also received unsolicited donations from its community members (people with arthritis) across Canada.
ACE thanks funders for their support to help the nearly 6 million Canadians living with osteoarthritis, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and the many other forms of the disease.
Disclaimer
The material contained on this website is provided for general information only. This website should not be relied on to suggest a course of treatment for a particular individual or as a substitute for consultation with qualified health professionals who are familiar with your individual medical needs. Should you have any healthcare related questions, you should contact your physician. You should never disregard medical advice or delay in seeking it because of something you have read on this or any website.
This site may provide links to other Internet sites only for the convenience of World Wide Web users. ACE is not responsible for the availability or content of these external sites, nor does ACE endorse, warrant or guarantee the products, services or information described or offered at these other Internet sites.
Although the information presented on this website is believed to be accurate at the time it is posted, this website could include inaccuracies, typographical errors or out-of-date information. This website may be changed at any time without prior notice.

Provincial and private drug benefit plans have begun implementing policy transition requiring patients to move from their current biologic originator to its biologic biosimilar. Policy transition has been successfully implemented in many European countries and in thousands of inflammatory arthritis patients with no compromise to patient safety, effectiveness or quality of care.
On May 27, 2019, British Columbia became the first jurisdiction in North America to introduce biosimilar policy transition with the launch of the Biosimilars Initiative, which will implement new reimbursement coverage for patients on infliximab or etanercept for rheumatoid arthritis, ankylosing spondylitis, and psoriatic arthritis.
Between May 27, 2019 and November 22, 2019, B.C. patients on Remicade® will be transitioned to its biosimilar, either infliximab-Inflectra® or infliximab-Renflexis®. Patients on Enbrel® will be transitioned to its biosimilar, either etanercept-Brenzys® or etanercept-Erelzi®. In each case, patients and their rheumatologist will be able to choose between the two available biosimilars.
Over the six month period, reimbursement coverage will be provided for all originator and biosimilar brands, in order to provide time for patients to speak with their rheumatologist about transitioning to a biosimilar.
Biosimilars are next generation biologic medications that have been approved for use in Canada since 2009, and in arthritis, since 2014. Fourteen biosimilars are currently approved by Health Canada with another 15 under review. Since the European Union (EU) approved the first biosimilar in 2006, the EU has approved 40 biosimilars.
In this special issue of JointHealth™ insight, we provide patients with inflammatory arthritis important biosimilars facts to help them understand the principles of this newest type of biologic and have an informed conversation with their healthcare provider.
What are biologic biosimilars?
 Biologics are life-saving “advanced therapy” treatments – meaning they are used after conventional disease modifying anti-rheumatic drugs (csDMARDs), such as methotrexate, hydroxychloroquine and sulfasalazine – that can help patients with inflammatory forms of arthritis. They are made from complex large molecules that come from living cells. Biologics are time consuming to research, develop and manufacture, making them expensive when they first come into the market place as a patented medicine. This can limit patients’ reimbursement access to such medicines and can make it difficult for the health care system to afford them.
Biologics are life-saving “advanced therapy” treatments – meaning they are used after conventional disease modifying anti-rheumatic drugs (csDMARDs), such as methotrexate, hydroxychloroquine and sulfasalazine – that can help patients with inflammatory forms of arthritis. They are made from complex large molecules that come from living cells. Biologics are time consuming to research, develop and manufacture, making them expensive when they first come into the market place as a patented medicine. This can limit patients’ reimbursement access to such medicines and can make it difficult for the health care system to afford them.Biosimilars are the next generation of biologic treatment and have nearly the same effectiveness, safety, immunogenicity profile and quality and deliver the same therapeutic benefits to patients as their originator; in some studies, slightly better.
After meeting Health Canada’s strict safety and efficacy requirements, biosimilars enter the market after a biologic originator patent expires, just like a small molecule generic medication does when the brand name medication’s patent expires. And just as generic medications are much lower in price compared to their original versions, so too are biosimilars.
For example:
- etanercept (Brenzys®) and etanercept (Erelzi®) are biologic biosimilar versions of the biologic originator etanercept (Enbrel®);
- infliximab-Inflectra® and infliximab-Renflexis® are biologic biosimilar versions of the biologic originator infliximab-Remicade®).
Biosimilars have been approved for use in Canada since 2009, and in inflammatory arthritis since 2014 – there are more biologic originators and biosimilars in use in inflammatory arthritis than in any other disease.
| The European Medicines Agency has published a guide to provide healthcare professionals with information on the science and regulation underpinning the use of biosimilars: “Over the last 10 years, the EU monitoring system for safety concerns has not identified any relevant difference in the nature, severity or frequency of adverse effects between 28 biosimilars and their reference medicines. |
Since the European Union (EU) approved the first biosimilar in 2006, it has approved the highest number of biosimilars worldwide, gaining experience of their use and safety. During this period, the EU monitoring system for safety concerns has not identified any relevant difference in the nature, severity or frequency of adverse effects between biosimilars and their biologic originators.
The science behind biosimilars
|
Biosimilars in Canada – approved and under review
| Biologic/ NBCD |
Molecule | Biosimilar | Indication | Notice of Compliance April 4, 2019 |
Biosimilar Manufacturer |
| Genotropin® | somatropin | Omnitrope | Growth hormone deficiency | 04/2009 | Sandoz |
| Remicade® | infliximab | Inflectra | AS, CD, PsO, PsA, RA, UC | 01/2014 | Hospira (Pfizer) |
| Remicade® | infliximab | Renflexis | AS, CD, PsO, PsA, RA, UC | 12/2017 | Samsung Bioepis (Merck) |
| Enbrel® | etanercept | Brenzys | AS, RA | 08/2016 | Samsung Bioepis (Merck) |
| Enbrel® | etanercept | Erelzi | AS, JIA, RA, PsA | 04/2017 | Sandoz |
| Humira® | adalimumab | Hadlima/Push Touch |
AS, CD, HS, PsO, JIA, PsA, RA, UC, Uveitis | 05/2018 | Samsung Bioepis (Merck) |
| Lantus/Solo Star® |
insulin glargine | Basaglar | Diabetes | 09/2015 | Lily |
| Lantus/Solo Star® |
insulin glargine | Semglee | Diabetes | N/A | BGP Pharma |
| Humalog® | insuline lispro | Admelog | Diabetes | 11/2017 | Sanofli |
What does transitioning mean?
| Transitioning (sometimes referred to as “switching”) generally refers to a one-time change from a biologic originator to a biologic biosimilar. | 
|
Will transitioned patients receive the same level of patient support from biosimilar manufacturers?
Patients should expect the same or similar types of support from all biologic patient support programs, no matter who makes their medication. The rheumatologist will give their patient the name of the patient support program coordinator for their biosimilar and they will help organize reimbursement and assist with other patient needs. The patient and their rheumatologist will monitor the safety and effectiveness of the biosimilar as part of routine patient care.
There are two types of transitions
 |
1 - “Medical transition” occurs when a person no longer responds and receives most or full benefit from the medication. Loss of response or “loss of efficacy” requires a person to change their medication(s). In the case of a patient not doing well on their biologic originator or biologic biosimilar, they would be medically transitioned either to a different biologic originator or biosimilar to regain maximum disease control. This is a carefully considered decision taken between the patient and their rheumatologist. Transitioning at the right time, when medically required, is important to achieving the best disease control and patient outcomes. |
 |
2 - “Policy transition” (sometimes referred to as “non-medical switching”) requires patients to move from their current biologic originator to its biologic biosimilar, usually because it is significantly less expensive. |
 |
What does Health Canada say about biosimilar transitioning?
|
The biosimilar transitioning experience
The leading regulators in the world – including the European Medicines Agency, Food & Drug Administration in the U.S and Health Canada – support well-controlled transition.
Patients need to know transition policy has been safely and effectively implemented over the past 10 years with thousands of patients with autoimmune diseases such as rheumatoid arthritis, psoriatic arthritis, gastrointestinal and bowel diseases in many countries in Europe with no compromise to patient safety, effectiveness or quality of care.
 More than 100 research studies exist looking at patients who have successfully policy transitioned from a TNF inhibitor biologic originator to its TNF inhibitor biologic biosimilar and show no health differences between patients.
More than 100 research studies exist looking at patients who have successfully policy transitioned from a TNF inhibitor biologic originator to its TNF inhibitor biologic biosimilar and show no health differences between patients.Today, the international inflammatory arthritis community has the most biosimilar experience of any disease community, with tens of thousands of patients safely and effectively transitioned to a biosimilar.
 |
“We’ve learned so much about the biosimilar transition experience from our fellow inflammatory arthritis patients in Europe – thousands have been transitioned to the newer version of their biologic and good information was key to their success. All biologics – originators and biosimilars – are made in batches, and after the first batch of the originator is gone, every batch thereafter becomes its biosimilar. For example, I have been on two biologic originators over the past 17 years, and I’ve been transitioned multiple times, from batch to batch to batch, and the benefits and risks to me have remained the same. The same is true for biologic biosimilars: batches are produced following strict guidelines set out by Health Canada. The only difference is biosimilars are produced off-patent and are roughly half the cost of the originator.” - Cheryl Koehn, Founder & President, Arthritis Consumer Experts |
Biosimilar Transitioning: The Canadian experience
In Canada, federal, provincial, territorial and private insurance drug plans have begun implementing policy transition, which moves patients from their current biologic originator to its biologic biosimilar. The European experience of rheumatologists successfully working with their patients to transition them from an originator to a biosimilar has helped inform policy transition development in Canada.
On May 27, 2019, British Columbia became the first jurisdiction in North America to introduce biosimilar policy transition with the launch of the Biosimilars Initiative. According to the Ministry of Health, the goal of the Biosimilars Inititiave is to improve the sustainability of its PharmaCare program by expanding the use of biosimilars, which will create opportunities for new drug listings and boost existing coverage for patients.
 |
“In my work leading Canada’s largest arthritis patient organization, I hear every day how patients like me are struggling to gain reimbursement for the medications their rheumatologist and patients feel they need. B.C. PharmaCare’s Biosimilars Initiative is a direct answer to this problem, clinically and financially. The initiative ensures continued reimbursement coverage for B.C. patients transitioned to the newer biologic, and ensures that reimbursement for other patients becomes available. This policy announcement shows how coverage for a few, turns into coverage for many. All of society benefits.” - Cheryl Koehn, Founder & President, Arthritis Consumer Experts |
Between May 27, 2019 and November 22, 2019, approximately 240,000 B.C. patients living with either ankylosing spondylitis, diabetes, plaque psoriasis, psoriatic arthritis or rheumatoid arthritis will have six months to work with their health-care team to transition their prescription from the biologic to the biosimilar drug by Nov. 25, 2019. After that time, PharmaCare will no longer provide coverage for the original biologic drug for these indications, except for exceptional cases.
An immediate effect of this announcement is the listing on BC PharmaCare of the psoriatic arthritis medication ixekizumab (Taltz®) as well as the diabetes medication empagliflozin (JARDIANCE™).
“B.C. is leading the country by promoting the widespread use of biosimilars, which have been proven to work just as safely and effectively as higher priced biologics. To date, Canada is far behind European jurisdictions,” said Adrian Dix, Minister of Health. “Biosimilars are a necessary step to ensure PharmaCare provides existing coverage for more people and funds new drugs well into the future.”
The Ministry of Health further explained that despite clear evidence to support the use of biosimilars – highly similar versions of biologics that cost anywhere from 25% to 50% less – Canada has a significantly lower use rate for biosimilars compared to other jurisdictions, at approximately 8%. Other nations have upwards of 50 to 95%.
Biologics, collectively, are the single biggest expense for public drug plans and their costs continue to climb. In 2018, B.C. spent $125 million on Lantus, Enbrel and Remicade, three biologic medications that treat chronic diseases such as diabetes, arthritis and Crohn’s disease.
“We know that our use rates can be much higher. For example, the biosimilars for infliximab (known as Inflectra® and Remsima®) have combined 5 to 10% use rate in Canada after four years on the market, compared to Norway (98%), United Kingdom (90%) and Scotland (85%). As a result, in 2017, Canada spent over $1.1 billion on the original biologic drug called Remicade®,” added Minister Dix.

Having an evidence-based biosimilar transition conversation with your healthcare professional
Canadian regulators recommend, prior to transition, a discussion occurs between a health care provider and their patient.
The most important element for patients going through a transition is access to patient-friendly, science-based information to support their conversations with their health care team.
Arthritis Consumer Experts (ACE) has been a leader in biosimilars discussions since 2009, sharing information with stakeholders across Canada through free research-based workshops, webinars and education programs. Drawing from this experience, ACE has created the Biosim•Exchange, an information hub for consumers to get the latest biosimilars news and background analysis. http://bit.ly/BiosimExchangeEN.
ACE has produced a special biosimilars education video series, where ACE speaks to medical experts on key patient questions around biosimilars and transitioning from originators to biosimilars: Go to https://biosim.jointhealth.org/resources and click on “Biosimilars education videos.”
ACE has also recently published a new guide -
| “Biosimilars in Canada: What inflammatory arthritis patients need to know” - to address those needs of patients who want information on biosimilar medicines. It aims to provide answers to questions patients may have on biosimilars and provide them the information tools they need to power and support their conversations with their rheumatologists and other health care providers and ensure science-based continuity of care. Click here to view the Guide: https://jointhealth.org/pdfs/BiosimilarsInCanadaGuide_EN.pdf. |
 |
Societal benefits of biosimilars
Why societal benefits are important to patients
Evidence-based private or public drug plan transition policy could help more patients (not just those to have been lucky enough to receive coverage for an originator under current and often restrictive special access criteria), the healthcare system, and society.

Societal benefit from biosimilar transition policy is not a “pipe dream” – it is a real opportunity to help more patients get on costeffective therapies that are also “state-of-the-art” in the treatment of inflammatory arthritis and other autoimmune diseases driven by inflammation.
Societal benefits to taxpayers
A key benefit of transitioning patients is the hundreds of millions of dollars in cost savings to the health care system.
In a presentation at a CADTH conference in April 2019, Elena Lungu from the Patented Medicine Prices Review Board (PMPRB) estimated the potential missed biosimilars savings in Canada for 2019 (versus the Organisation for Economic Cooperation and Development (OECD) countries with the highest biosimilars uptake are achieving):
| Infliximab | $482 M | = $791 M |
| Etanercept | $140 M | |
| Insulin | $124 M | |
| Filgrastim | $45 M |
That translates into a total of $791 million of biosimilars savings in 2019 potentially not realized by Canadian public and private drug plans.
A study presented at the 2019 Canadian Rheumatology Association annual meeting looked at biosimilar use and cost in Canada under provincial drug plans in 2016 (excluding Quebec) and found that if half of the Remicade claims has been Inflectra instead, the cost difference would have been more than $102 million1.
According to a 2018 study commissioned by the PMPRB and conducted by the National Prescription Drug Utilization Information System (NPDUIS), “Potential Savings from Biosimilars in Canada”, estimated potential annual savings for a single biologic originator, like infliximab, in Canada could range between $91M and $514M in the third year following biosimilar entry.
Evidence-based provincial drug plan and health insurer transition policy could help more patients, the healthcare system, and society, if savings from biosimilars use was redirected to:
- Facilitate revision of “special access criteria” to ensure patients have access to the right drug at the right time
- Re-invest in formulary budgets (e.g. adding new medications and improving outcomes for more patients)
- Invest in non-medication types of care that patients need, such as specialized nursing, physio- and occupational therapy, and other important elements
- Extend life-time drug benefit (60% of prescriptions are paid for by employer provided drug benefits; most have life-time limits for biologic reimbursement)
| Real and potential benefits from biosimilars savings | ||
| Benefit | Originators | Biosimilars |
| Safe and effective (Health Canada approved) |  |
 |
| Innovative |  |
 |
| Savings to taxpayers |  |
|
| Policy drivers |  |
|
| Formulary modernization |  |
|
| Potential to advance models of care in chronic disease |  |
|
| Please share this JointHealth™ insight with other patients, your friends and family. |  |
| If you have any questions or comments, we want to hear from you. |  |
Listening to you
We hope you find this information of use. Please tell us what you think by writing to us or emailing us at feedback@jointhealth.org. Through your ongoing and active participation, ACE can make its work more relevant to all Canadians living with arthritis.
Update your email or postal address
Please let us know of any changes by contacting ACE at feedback@jointhealth.org. This will ensure that you continue to receive your free email or print copy of JointHealth™ insight.
Arthritis Consumer Experts (ACE)
Who We Are
Arthritis Consumer Experts (ACE) operates as a non-profit and provides free research based education and information to Canadians with arthritis. We help (em)power people living with all forms of arthritis to take control of their disease and to take action in healthcare and research decision making. ACE activities are guided by its members and led by people with arthritis, scientific and medical experts on the ACE Advisory Board. To learn more about ACE, visit www.jointhealth.org
Disclosures
Over the past 12 months, ACE received grants-in-aid from: Amgen, Arthritis Research Canada, Canadian Biosimilars Forum, Canadian Institutes of Health Research, Canadian Rheumatology Association, Eli Lilly Canada, Hoffman-La Roche Canada Ltd., Knowledge Translation Canada, Merck Canada, Novartis Canada, Pfizer Canada, Sandoz Canada, Sanofi Canada, UCB Canada, and the University of British Columbia.
ACE also received unsolicited donations from its community members (people with arthritis) across Canada.
ACE thanks funders for their support to help the nearly 6 million Canadians living with osteoarthritis, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and the many other forms of the disease.
Disclaimer
The material contained on this website is provided for general information only. This website should not be relied on to suggest a course of treatment for a particular individual or as a substitute for consultation with qualified health professionals who are familiar with your individual medical needs. Should you have any healthcare related questions, you should contact your physician. You should never disregard medical advice or delay in seeking it because of something you have read on this or any website.
This site may provide links to other Internet sites only for the convenience of World Wide Web users. ACE is not responsible for the availability or content of these external sites, nor does ACE endorse, warrant or guarantee the products, services or information described or offered at these other Internet sites.
Although the information presented on this website is believed to be accurate at the time it is posted, this website could include inaccuracies, typographical errors or out-of-date information. This website may be changed at any time without prior notice.
